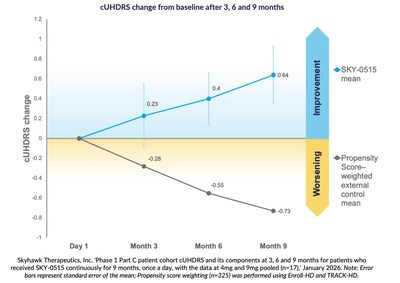
Nine-month results show an average improvement in the Unified Composite Huntington’s Disease Rating Scale from baseline of +0.64 points., Compared to natural history, cUHDRS in symptomatic patients is expected to worsen by -0.73 points over nine months, based on propensity score weighting.
Skyhawk also announces that the Phase 2/3 FALCON-HD trial of SKY-0515 has expanded globally. Skyhawk has now administered doses to more than 90 patients.
BOSTON, January 27, 2026 /PRNewswire/ — Skyhawk Therapeutics, Inc.a clinical-stage biotechnology company developing novel small molecule therapeutics to modulate critical RNA targets, today announces positive results from the nine-month interim analysis of the Company’s investigational treatment for Huntington’s disease (HD) with SKY-0515.
Treatment with SKY-0515 results in a dose-dependent reduction in blood mHTT protein by 62% at the 9 mg dose and a dose-dependent reduction in PMS1 mRNA by 26%. PMS1 is a key driver of somatic CAG repeat expansion and HD pathology. SKY-0515 also demonstrated excellent central nervous system exposure and was generally safe and well tolerated.
At three, six and nine months, patients receiving SKY-0515 in the Part C patient cohort of the SKY-0515 Phase 1 clinical trial demonstrate a mean improvement in the Unified Composite Huntington’s Disease Rating Scale (cUHDRS) compared to baseline. At nine months, in a pooled analysis, this improvement is +0.64 points compared to an expected nine-month worsening of cUHDRS in symptomatic patients of -0.73 points, based on propensity score weighting using Enroll-HD and TRACK-HD.
“I am very encouraged by these early safety and efficacy data from the Phase 1 Part C trial of SKY-0515 in patients, showing divergence of cUHDRS from expected natural history deterioration at prespecified three-, six- and nine-month analyses,” said Ed Wild, professor of neurology at University College London. “SKY-0515 continues to reduce mHTT protein to the greatest extent demonstrated by any treatment tested to date in patients, with clinical and biomarker data showing the drug is well tolerated at all doses tested. The ability of SKY-0515 to reduce both mHTT and PMS1 provides a potent combination to treat Huntington’s disease via two of its major pathogenic mechanisms. a significant impact is expected for people living with HD across the world – for whom an orally administered huntingtin-lowering treatment such as SKY-0515 will be truly transformative.
“Our goal for our Phase 1 study was to establish the safety and activity of the biomarkers,” said Sergey Paushkin, head of R&D at Skyhawk Therapeutics, “and the continued strength of the biomarker response of SKY-0515 during our nine-month interim analysis – as well as the improvement in the potential endpoint, cUHDRS, compared to a worsening of the cUHDRS score in the natural history data of the patients – highlight the potential of SKY-0515 as a best-in-class disease-modifying therapy for HD. These interim data represent an important milestone for SKY-0515 and highlight the power of Skyhawk’s platform to deliver first-in-class small molecules for devastating diseases without approved disease-modifying therapies.
Huntington’s disease is a rare, inherited, and ultimately fatal neurodegenerative disorder that affects more than 40,000 symptomatic patients in the United States, with hundreds of thousands estimated to be affected worldwide. There are currently no approved treatments to slow or stop the progression of the disease. SKY-0515 is an investigational, orally administered, small molecule RNA modulator developed through the Company’s novel RNA modulation platform, SKYSTAR.®. SKY-0515 therapeutically reduced both HTT protein and PMS1 protein. PMS1 is an additional key driver of somatic CAG repeat expansion and HD pathology and is expected to complement the benefits of reduced mutant HTT.
Skyhawk also announces today that its Phase 2/3 FALCON-HD trial SKY-0515, opened at twelve sites in Australia and New Zealand, has expanded globally. Skyhawk has now administered SKY-0515 to over 90 patients.
SKY-0515 is the first Skyhawk drug in clinical trials.
Skyhawk plans to bring additional small molecule drugs to the clinic to treat rare neurological diseases without approved disease-modifying therapies by the end of 2027.
About the Phase 1 clinical study of SKY-0515
The Phase 1 clinical trial of SKY-0515 is the first-in-human trial designed to evaluate the safety, tolerability, pharmacokinetics and pharmacodynamics of SKY-0515 in healthy volunteers and individuals with early-stage Huntington’s disease (HD). The trial is divided into three parts. Parts A and B evaluated SKY-0515 in healthy volunteers. Part C is a double-blind, placebo-controlled parallel study of two dose levels of SKY-0515 and placebo in individuals with early-stage HD (HD-ISS Stage 1, 2, or Light Stage 3) for 84 days followed by a 12-month extension of active treatment during which all participants will receive a low or high dose of SKY-0515 in a blinded manner. Study objectives include evaluation of mutant HTT protein and PMS1 mRNA. The first patients received treatment in Part C of SKY-0515 in January 2025. Enrollment for Phase 1C of the SKY-0515 trial is now complete.
About the Phase 2/3 FALCON-HD clinical study of SKY-0515
FALCON-HD (NCT06873334) is a phase 2/3 randomized, double-blind, placebo-controlled study to evaluate the pharmacodynamics, safety and efficacy of SKY-0515 in 120 participants with stage 2 and early stage 3 HD at 12 sites in Australia and New Zealand, and 400 participants with stage 2 and early stage 3 HD at over 40 sites worldwide. Eligible patients will receive a once-daily oral dose of SKY-0515 at one of three dose levels or placebo, for a treatment period of at least 12 months. The trial aims to evaluate the potential of SKY-0515 to modulate RNA splicing and reduce mHTT and PMS1 proteins, which are involved in the pathology of Huntington’s disease. Additional information about FALCON-HD, including participating sites and eligibility criteria, is available at ClinicalTrials.gov And www.FALCON-HD.com.
About Skyhawk Therapeutics
Skyhawk Therapeutics is a clinical-stage biotechnology company utilizing its proprietary platform, SKYSTAR®to discover and develop small-molecule RNA-modulating therapies for the world’s most incurable diseases. For more information, visit www.skyhawktx.com.
Contact Skyhawk
Maura McCarthy
Corporate development manager
maura@skyhawktx.com

SOURCE Skyhawk Therapeutics